DPhil Student
Nuffield Department of Surgical Science
University of Oxford
A spatially resolved analysis of immune responses within rejecting skin grafts from humanised mice, with and without low dose Treg treatment
Sarah Short1, Amy Cross 1, Joanna Hester1, Fadi Issa1.
1Nuffield Department of Surgical Sciences, University of Oxford , Oxford, United Kingdom
Translational Research Immunology Group (TRIG).
Introduction: Skin transplant rejection is a significant problem in vascularised composite allotransplantation. To address this, several therapeutic approaches are being developed, including cellular therapy with polyclonal regulatory T cells (Tregs). Understanding the mechanisms that are relevant to skin susceptibility to rejection and the immune regulatory mechanisms that may be promoted by Treg treatment is crucial. In this setting, the ratio of effector to Tregs determines graft survival outcome, with high doses of Tregs resulting in potent systemic immune suppression. Spatial transcriptomic techniques have emerged as a valuable tool to understand cell phenotypes and interactions within tissue, while retaining anatomical architecture that may be mapped back to Banff classification. In this study we aimed to assess the character of immune responses within skin grafts from humanised mice transplanted with human skin, with and without Treg treatment at low cell doses, to help identify the early tissue immune regulatory signatures that may be emerging prior to more systemic immune suppression.
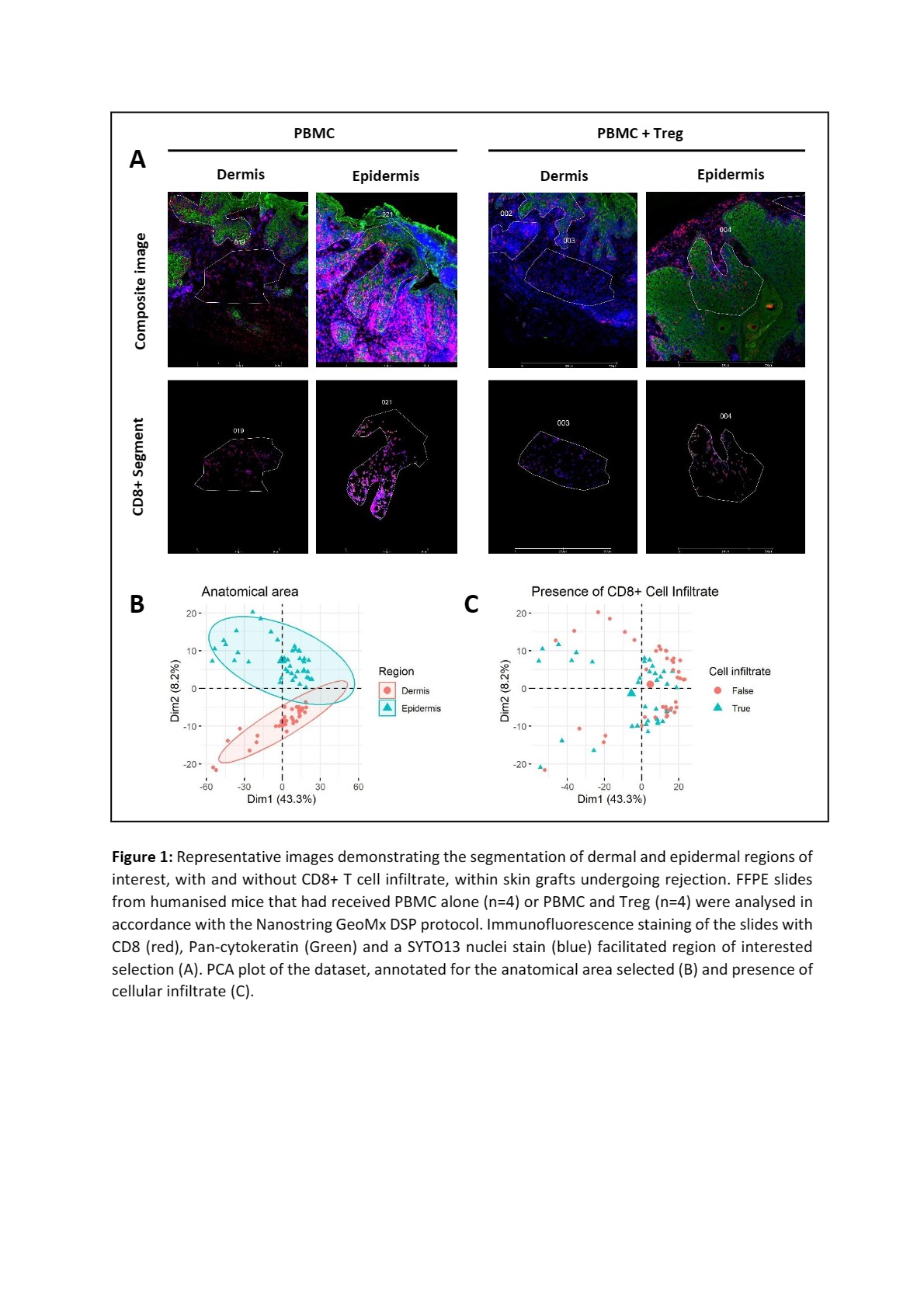
Methods: An analysis of rejecting skin allografts from a humanised mouse model was completed to define the tissue signatures of rejection and regulation within the allograft. Immunodeficient BALB/c BRG mice received human skin grafts prior to intraperitoneal PBMC alone (n=4) or PBMC and Treg (n=4) at a ratio of 5:1. Control mice received a skin graft alone (n=3). Harvested formalin fixed paraffin embedded (FFPE) skin grafts were analysed according to the Nanostring GeoMx Digital Spatial Profiling (DSP) workflow using the whole transcriptome atlas and segmented based on areas of dermis and epidermis with and without CD8+ cell infiltrate (Figure 1). FFPE slides were H&E stained for rejection grading and immunohistochemical staining for FOXP3 completed. Splenocytes from the rejecting mice were analysed by flow cytometry.
Results: Low-dose Treg transfer did not significantly alter rejection outcomes, determined by Banff rejection grade at endpoint. Consistent with this outcome, Treg frequency and quality did not significantly differ between the groups on histological and flow cytometric analysis (p >0.05, unpaired t test). Numerous immune related genes, including several unexpected immunoglobulin specific genes, were upregulated in the immune infiltrate of PBMC treated skin grafts compared to control. A limited number of genes were differentially expressed on analysis of the immune infiltrate between treatment groups, including TSPAN32, MSRB2 and RPL19. Conclusion: Although low dose Treg therapy did not significantly alter rejection outcomes, this dataset may provide insights into the early tissue specific features of immune regulation.
Lectures by Sarah M Short
| When | Session | Talk Title | Room |
|---|---|---|---|
|
Tue-02 09:20 - 10:30 |
Abstracts Session 3 | A spatially resolved analysis of immune responses within rejecting skin grafts from humanised mice, with and without low dose Treg treatment | Grand Georgian |
