
Dr. Lori West is a Professor of Pediatrics, Surgery, Medical Microbiology/Immunology and Laboratory Medicine/Pathology at the University of Alberta and Director of the Alberta Transplant Institute. A clinician-scientist, she has longstanding interest and expertise in pediatric heart transplantation and transplant immunology, particularly related to ABO glycoimmunology. Her pioneering work on crossing ABO-barriers led to global impact on infant heart transplantation. Dr. West is also the Director of the Canadian Donation Transplant Research Program, a national research coalition funded since 2013 by the Canadian Institutes of Health Research. She previously held the Tier 1 Canada Research Chair in Heart Transplantation, is past-president of the International Society of Heart and Lung Transplantation and the Canadian Society of Transplantation, and immediate past-chair of the Women in Transplantation international initiative of The Transplantation Society. A Fellow of both the Royal Society of Canada and the Canadian Academy of Health Sciences, Dr. West was appointed as an Officer of the Order of Canada in 2020.
Enzymatic conversion of blood group A- to H-antigen in a mouse model to facilitate ABO-incompatible transplantation (ABOi Tx)
Tate Erickson1, Bruce Motyka1, Lai Xu1, Jean Pearcey1, Marcelo Cypel2, Jayachandran Kizhakkedathu3, Peter Rahfeld4, Peter J Cowan5, Stephen G Withers6, Lori J West1.
1Pediatrics, University of Alberta, Edmonton, AB, Canada; 2Surgery, University of Toronto, Toronto, ON, Canada; 3Pathology and Laboratory Medicine, University of British Columbia, Vancouver, BC, Canada; 4Avivo Biomedical Inc., Vancouver, BC, Canada; 5Immunology Research Centre, St Vincent’s Hospital Melbourne, Melbourne, Australia; 6Chemistry, University of British Columbia, Vancouver, BC, Canada
Purpose: The ABO histo-blood group system presents a major barrier in organ Tx that limits organ availability. A reduction in donor A/B antigens represents a novel approach that may allow safe ABO-Incompatible Transplantation (ABOi Tx). We recently demonstrated the utility of FpDeAc (Deacetylase) and FpGalNase (Galactosaminidase), called Azymes, to convert blood group A to blood group O in an ex vivo human lung perfusion model, however, the duration of A-antigen removal remains unclear. A-transgenic (Tg) mice developed by our group express A-antigen on vascular endothelium and red blood cells (RBC) and have been used to model ABOi Tx. Using this model, we assessed A-antigen removal and re-expression following Azymes administration in vitro and in vivo.
Methods: Azymes function was studied using A-Tg BALB/c (male/female, n=5/3) and A-Tg C57BL/6 (male/female, n=2/4) mice (10-44 wk). In vitro: RBC were assessed for A- and H-antigen expression by hemagglutination at various timepoints (0.5-4 hr) post-Azymes treatment (2-50 µg/mL) of whole blood. In vivo: A-Tg mice were injected iv with Azymes (0.4-0.8 mg/kg); blood was sampled at various timepoints (0.25-96 hr) and RBC were tested for A- and H-antigen expression by flow cytometry and hemagglutination. Heart and lung tissue were assessed at various times (0.5-96 hr) for A- and H-antigen expression by immunohistochemistry.
Results: In vitro: Azymes fully converted A- to H-antigen on A-Tg mouse RBC (Fig. 1). In vivo: Azymes treatment fully converted A- to H-antigen on A-Tg mouse RBC, persisting up to 2 hr post-treatment. Heart and lung vascular endothelium showed a marked reduction in A-antigen and conversion to H-antigen up to 8 hr post-treatment (Fig. 2). No significant sex differences were observed.
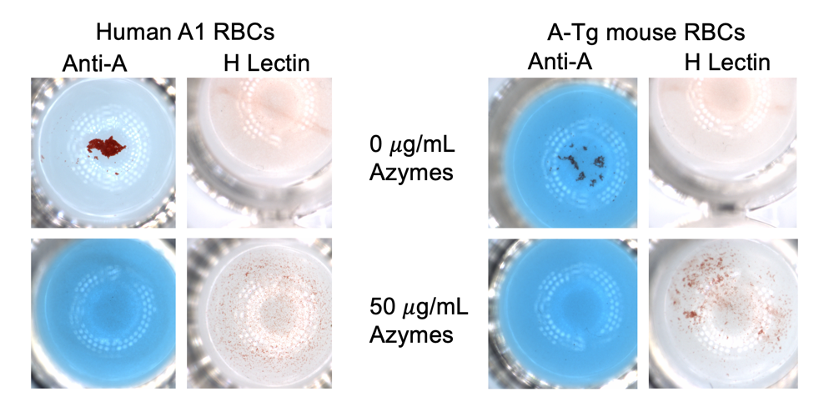
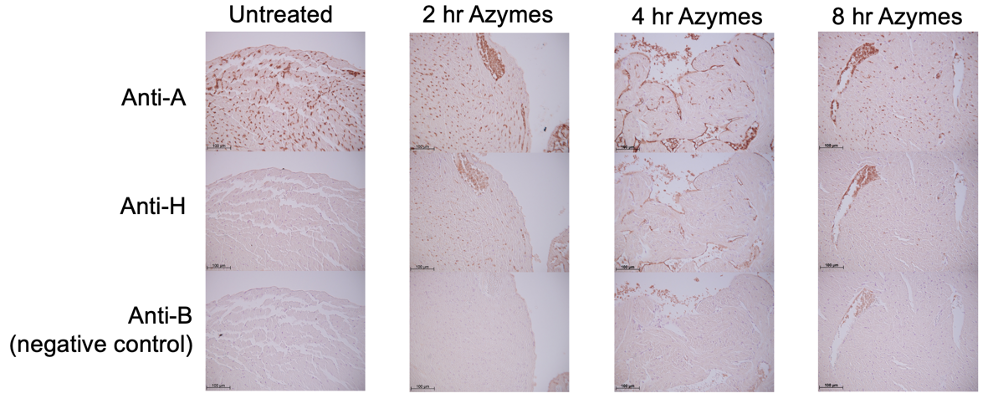
Conclusions: Our findings confirm that Azymes effectively remove A-antigen from RBC and heart and lung tissues in mice, and show decreased tissue expression persisting for at least several hours. Future studies will assess the ability of Azymes treatment to prevent antibody-mediated rejection in the A-Tg mouse model of ABOi Tx. Clinical application of Azymes technology has the potential to expand ABOi organ Tx, allowing lifesaving Tx in individuals for whom compatible organs cannot be found, and use of donated organs otherwise discarded due to lack of compatible recipients.