CD38 activation with methionine choline deficient (MCD) diet-induced liver injury in rats
Jung Lye Kim1,2, Yong Gyu Lee1,2, Joanna M. Marshall1,2, Bryan A. Whitson1,2, Sylvester M. Black1,2.
1The COPPER Laboratory, The Ohio State University Wexner Medical Center, Columbus, OH, United States; 2Department of Surgery, The Ohio State University Wexner Medical Center, Columbus, OH, United States
Background: Nonalcoholic fatty liver disease (NAFLD) is the most common liver disorder in westernized countries, and a key factor impacting the quality of donor organs available for transplantation. In recent years, extended criteria donor (ECD) grafts have become more common, resulting in transplantation of organs impacted by metabolic conditions such as NAFLD or non-alcoholic steatohepatitis (NASH). Therefore, effective treatment of conditions such as steatosis, fibrosis and inflammation hold the potential to improve patient care at two critical levels: first, by improved treatment of the initial disease, and second, by protecting/increasing donor organ quality and function. We have identified that CD38, a multifunctional ectoenzyme and modulator of intracellular calcium metabolism, contributes to the inflammation associated with fatty livers and may serve as a therapeutic target. Here, we investigate the role of CD38 in a methionine/choline-deficient (MCD) fatty liver rat model.
Method: Animals were fed either an MCD diet, or standard diet, for 4 weeks. Development of fatty liver was confirmed by histology and biochemical analysis. Liver tissue was adebosine triphospage (ATP) and NAD/NADH ratio. Primary cells were isolated from either MCD-fed or standard-diet livers and evaluated for CD38 expression and activity in each of the major liver cell types.
Results: Animals fed the MCD diet developed fatty liver with characteristic markers of organ injury, inflammation, fibrosis, oxidative stress and metabolic disequilibrium. The liver tissue of MCD-fed animals had lower NAD/NADH ratios and reduced ATP, consistent with a depletion of cellular NAD pools. Analysis of isolated primary cells revealed that the MCD-fed animals displayed increased CD38 expression and activity in the liver, specifically on the surface of Hepatocytes, while expression in other cell types remained relatively constant. This increased expression was largely driven by a sub-population of small hepatocytes, which was significantly increased by the MCD diet.

Figure 1. SD rats were fed for 4 weeks on Normal diet or MCD diet. NAD content (A) and NADH (B) content were determined in the liver tissues. NAD/NADH ratio was determined as the total NAD content divided by total NADH levels (C). ATP levels were determined in the liver tissue
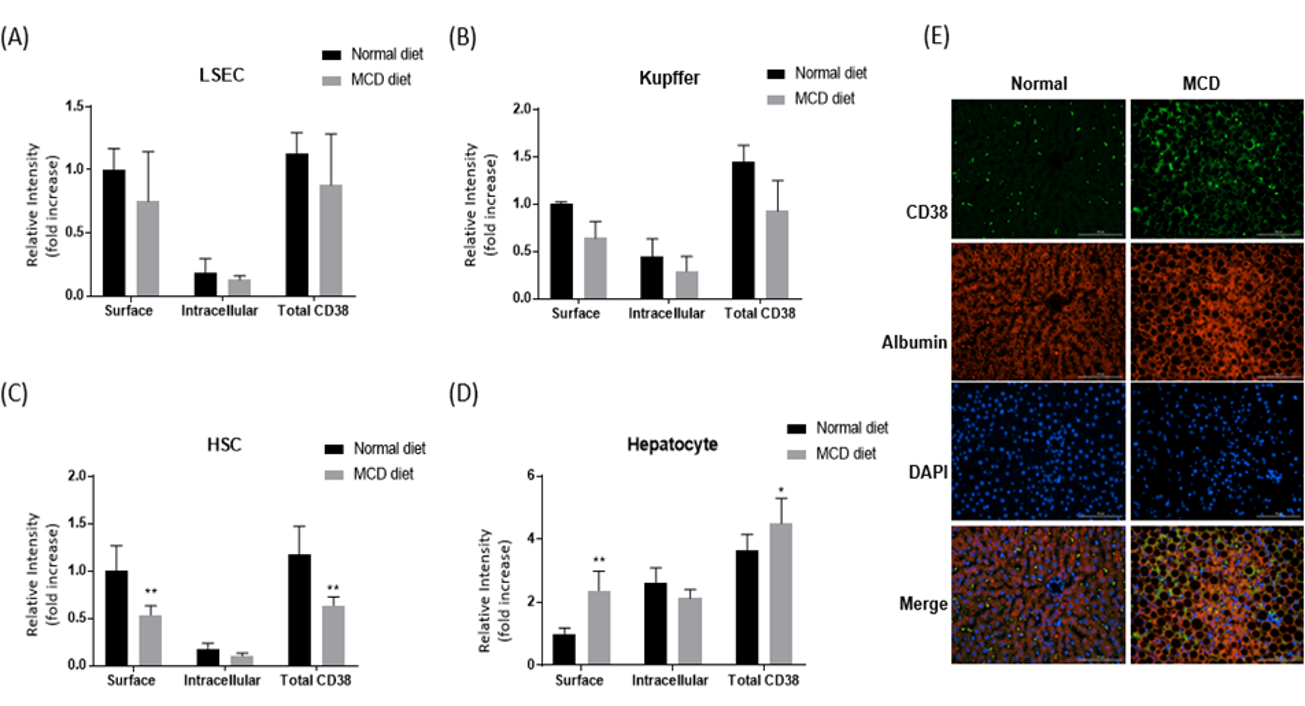
Figure 2. The CD38 expression and location in each cell type in normal and MCD diet for 4 weeks. The Liver sinusoidal endothelial cells (A), Kupffer cells (B), Hepatic stellate cells (C) and Hepatocyte (D). isolated from normal and MCD diet and stained with CD38 in surface and intracellular. The liver sections of normal and MCD diet were stained CD38 and Albumin (E).
Conclusion: Taken together, these data suggest that CD38 contributes to the pathophysiology of steatohepatosis-mediated liver injury through cellular damage, inflammation, depletion of NAD pools. We identified a population of small hepatocytes with high CD38 expression and activity, and which is specifically increased in animals fed the MCD diet. Future studies will explore the role of this small hepatocyte population in liver inflammation, fibrosis and repair.
[1] Karim Gariani. Inhibiting poly ADP-ribosylation increases fatty acid oxidation and protects against fatty liver disease. J Hepatol. 2017; 66(1): 132-141.
[2] Hiroko Itagaki. Morphological and functional characterization of non-alcoholic fatty liver disease induced by a methionine-choline-deficient diet in C57BL/6 mice. Int J Clin Exp Pathol. 2013; 6(12): 2683-2696.
[3] Frances E Lund. Signaling Properties of CD38 in the Mouse Immune System: Enzyme-dependent and -independent Roles in Immunity. Molecular Medicine. 2006; 12: 328-333.