Conversion to Belatacept in Kidney Transplant Recipients
Mahmoudreza Moein1, Benson Li1, Peter Fioramonti 1, Rauf Shahbazov1, Samir Iskhagi1, Matthew Hanlon1, Reeba Thankachan1, Christine Yang1, Reza Saidi1.
1Surgery/ Transplantation services, SUNY Upstate Medical University, Syracuse, NY, United States
Background: The costimulatory inhibitor Belatacept (Bela) has been shown to be an effective alternative as a post-kidney transplant immunosuppressant. This study focused on the outcome of early vs. late conversion to the Belatacept.
Material and Method: We conducted a retrospective analysis of a prospectively collected database of all kidney transplants adult patients at SUNY Upstate Medical Hospital from 1 January 2014 to 1 December 2022. . The early conversion was defined as all conversions done at < 6 months after kidney transplant, and Late conversion to Belatacept was defined as conversion at > 6 months post-transplantation.
Results: 61 patients were included in the study and divided into early and late conversion to Belatacept groups. 33 patients (54%) were in the early conversion group, and 28 patients (46%) were in the late conversion group. The mean eGFR was 26.73 before conversion to the Belatacept in the early conversion group, which improved over one year of follow-up with the mean eGFR of 45.3 at one year (P=0.0006). eGFR changes were insignificant in the late conversion group, in which the mean eGFR was 46.30 before conversion to the Belatacept, and it was 44.76 after one year of follow-up and stabilized (P=0.72).
7 allograft biopsy-proven rejection episodes were diagnosed overall, 4 episodes (57.1%) in the early conversion group and 3 episodes (42.9%) in the late conversion group. In 6 episodes (85.7%), patients did not have Tacrolimus in their immunosuppression regimen with Belatacept. However, when we analyzed the whole sample, we did not mention a significant difference in the rejection rate when Tacrolimus was part of the immunosuppressive regimen (P=0.6). All 4 biopsy-proven allograft rejections in the early conversion group were acute T-cell-mediated rejections. In the late conversion group, from 3 biopsy-proven rejections, 1 was chronic antibody-mediated rejection (CAMR), 1 T-cell-mediated rejection, and 1 was mixed antibody-mediated and T-cell-mediated rejection.
Surprisingly, in all 4 T-cell-mediated rejections (100%) in the early conversion group and, the one T-cell-mediated rejection in the late conversion group, mycophenolic acid (MPA) was part of the patient’s immunosuppressive regimen.
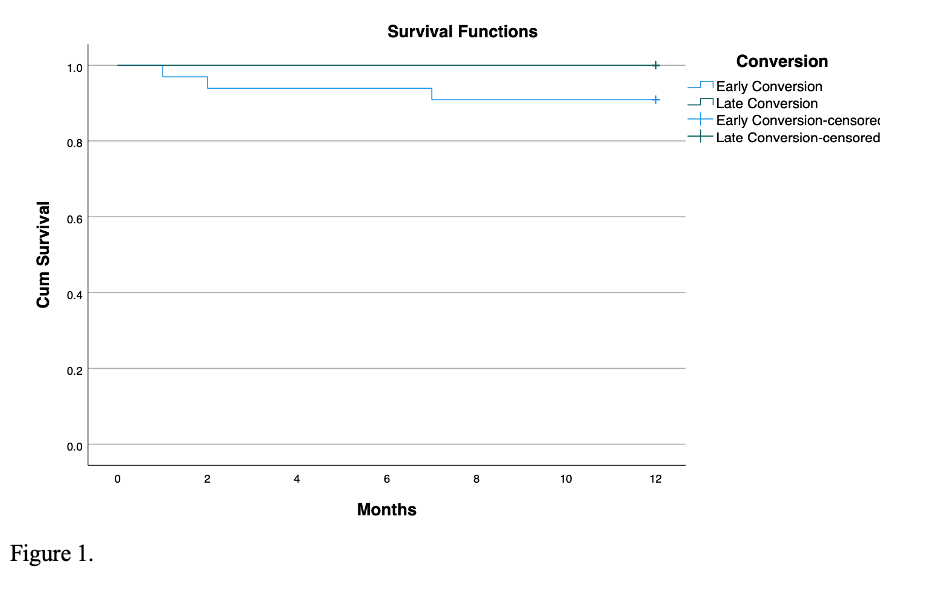
The one-year post-conversion allograft survival rate in both groups was 100%. The one-year post-conversion patient survival rate was 90.9% in the early conversion group and 100% in the late conversion group (P=0.11, Fig 1).
Conclusions: We conclude that early conversion to the Belatacept can significantly improve the eGFR compared to the late conversion. Belatacept and MPA without Tacrolimus might increase the chance of T-cell-mediated rejection.